Physically synthesized Ni-Cu
nanoparticles for magnetic hyperthermia
Martin Bettge, Jhunu Chatterjee and Yousef Haik*
Address: Center for Nanomagnetics and
Biotechnology Florida State University Tallahassee, Florida 32310 USA
Email: Martin Bettge - bettge@eng.fsu.edu; Jhunu
Chatterjee - jhunu@eng.fsu.edu; Yousef Haik* - haik@eng.fsu.edu
* Corresponding author
Abstract
Background: In this paper, a physical method to
prepare copper-nickel alloy particles in the sub-micron range for possible self
controlled magnetic hyperthermia treatment of cancer is described. It is
reported that an increase in tumor temperature decreases the tumor resistance
to chemo-and radiation therapies. Self controlled heating at the tumor site to
avoid spot heating is managed by controlling the Curie temperature of the
magnetic particles. The process described in this paper to produce the
nanomagnetic particles allows for a large scale production of these particles.
Methods: The process used here is mainly composed of
melting of the Cu-Ni mixture and ball milling of the resulted bulk alloy. Both
mechanical abrasion and continuous grinding were used to break down the bulk
amount into the desired particle size.
Results: It was found that the desired alloy is
composed of 71 % nickel and 29% copper by weight. It was observed that the
coarse sand-grinded powder has a Curie temperature of 345 K and the fine
ball-milled powder shows a temperature of 319 K - 320 K.
Conclusion: Self regulating magnetic hyperthermia can
be achieved by synthesizing nanomagnetic particles with desired Curie
temperature. In this study the desired range of Curie temperatures was obtained
by combination of melting and ball milling of nickel-copper alloy.
Background
Localized hyperthermia technique using magnetic particles,
based on proposal brought forward by Gilchrist in 1957, continues to be an
active area of research. It has been found that the viability of cancer cells
is reduced and their sensitivity to chemotherapy and radiation increase when
the human or animal malignant cells are heated to temperatures between 41-46°C
[1-3]. Magnetic hyperthermia provides the heat at the site of the tumor
inva-sively by applying an external alternating magnetic field to the magnetic
particles at the tumor site. The particles will heat up and conduct the heat to
the tumor cells. The use of materials with Curie temperature in the range of
41-46 ° C is desired to provide a safeguard against overheating of normal
cells, due to the decrease of magnetic coupling in the paramagnetic regime
(above Tc). The binary alloy copper-nickel shows a promising magnetic phase
transitions in the desired range of temperature for hyperthermia treatment of
cancer.
The phase equilibria system for copper-nickel shows a
linear progression for the Curie temperature, which starts at a composition of
67% nickel and 33% copper (by weight) for a temperature of 0°C [4]. From the
phase diagram of Cu-Ni alloy, the optimum amount of nickel in the alloy is
determined to be 71-71.4% by weight to have a Curie temperature in the desired
range of 41-46 °C. A Cu-Ni alloy for hyperthermia applications has been
produced first by Lilly et al [5]. They fabricated self-regulating implants via
physical melting. Bimetallic nanoparticles can be synthesized by a wide variety
of physical methods, such as, sputtering [6], mechanical alloying (ball
milling) [7,8], eletrodeposition [9] or partial recrystallization of amorphous
materials [10]. Most of the methods yield two-phase nanocrystalline materials.
For instance, Guo et al. [9] have produced composite Cu-Ni nanostructures via
an electrodeposition and template-based process. Natter et al. [11] used a
pulsed electrodeposition process, enabling the control of grain size and
chemical composition of the deposited material. Control of the composition in
the nanolevel is difficult [12], since molecules and atoms in common techniques
(e.g. chemical vapor deposition, plasma vapor deposition) do not necessarily
arrange in the preferred composition, which was determined on bulk material on the
macroscopic level.
In the present study we used a simple process that combines
melting and ball milling of bulk materials. Koch [13] has reviewed the facts of
ball milling (mechanical alloying) and its impact on nanostructured materials,
indicating that ball milling can produce average grain sizes below 100 nm. Even
though Natter [11] has shown that the chemical composition can be controlled
via pulsed electrodeposition; and deposition on a porous substrate could
possibly yield nanoparticles, it is nevertheless a complex process requiring
expensive equipment, the control of several parameters and a lot experience.
Additionally, this process is not feasible for
industrial high-scale applications.
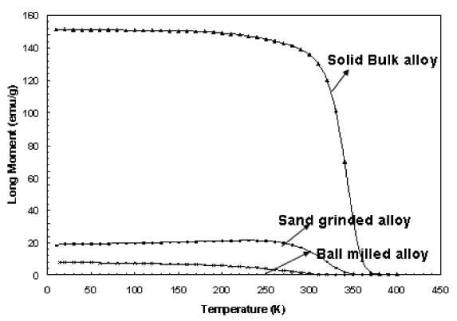
Figure 1
Magnetization and magnetic phase transition
(Curie temperature) of the as-produced, sand grinded and ball-milled
copper-nickel alloy (29% wt. copper, 71% wt nickel), from top to bottom,
respectively.

Figure 2
Electron diffraction pattern for ball milled
Cu-Ni particles.
Table 1: Comparison of XRD on sand grinded powder and
Electron Diffraction on ball-milled powder.
|
XRD |
2 θ angle (°) |
d-spacing (Angstrom) |
|
(1 1 1) (1stpeak) |
44.24 |
2.045 |
|
Electron Diffraction (111) Inner ring |
44.44 |
2.036 |
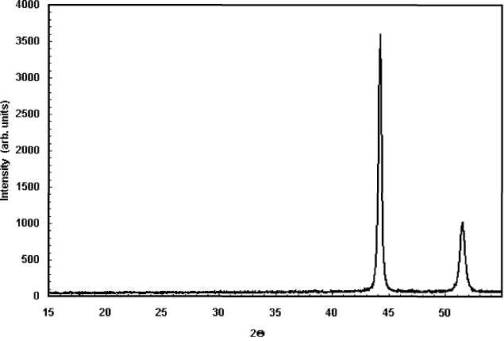
Figure 3
Wide angle X-Ray diffractogram for sand
grinded Cu-Ni particles.
Materials and methods
The nickel-copper alloy was obtained via physical melting,
in which nickel powder (AlfaAesar, 325 mesh, 99%) and copper powder (AlfaAesar,
500 mesh, 99%) were mixed in the desired composition (71% nickel, 29% copper;
w/w). In order to obtain a highly homogenous composition over the resulting
bulk alloy, the mixture was ball milled for 2 hours, before it was placed into
an alumina crucible. The mixture was heated up to 1465 °C for 3 hours under
nitrogen (prevention of oxidation). The liquid temperature of the alloy is
1365 °C [4] - a higher temperature was used in order to avoid inaccuracies due
to differences between actual and set temperature.
Basically, two steps are necessary to break down the
bulk chunk to the desired particle size. The first one is done by any
mechanical abrasion for producing a powder texture that enables the subsequent
use of a continuous grinding media as a second step. This effort made use of
simple and automated grinding at first and additional grinding up in a ceramic
ball mill for at least 3 to 7 days. It was carried out in a wet environment
using acetone in order to enhance the mixing of the content that is being
milled and to prevent oxidation and the development of a metallic particle gas
that is toxic and not collectible. Ten ceramic (alumina) balls were used for 5
g of starting material. The ball weight to content weight ratio was 6. The
rotation speed of a jar (40 mm in diameter) was 120 min-1. After
decanting from the jar, the dispersion was dried in vacuum. A highly dispersed
grayish-metallic suspension was obtained after the first day of ball milling.
A JEOL 2010 transmission electron microscope was used
to determine the particle morphology. A ZetaPALS Particle Size Analyzer
(Brookhaven Instrument Corp.) was used to determine the particle size. Magnetic
properties were measured using an MPMS 5 SQUID magnetometer.
Wide angle X-ray diffraction pattern was taken in a
Siemens 500 X-ray diffractogram with CuKα (λ = 0.154 nm)
radiation.
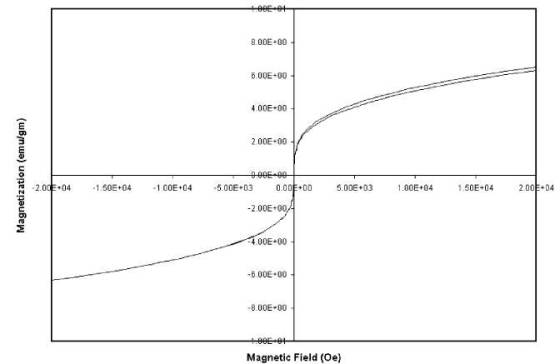
Figure 4
Hysteresis plot of the
ball-milled powder at 300 K. Magnetic behavior is similar to that of
superparamagnetic materials.
Results and discussion
Ni-Cu alloy is a relatively soft material (Rockwell
hardness of B25 [14] for a Ni-Cu 70:30 alloy) compared to a Rockwell hardness
of N80 for alumina [14], offering the possibility for further mechanical
treatments in order to achieve desired particle sizes. The size of the
particles is restricted by the smallest diameter of human blood vessels to
approximately 400 nm.
Figure 1 shows the magnetization of the as-produced
bulk alloy as well as the grinded and ball-milled powder in a magnetic field of
100 Oe. It indicates for the as-produced bulk alloy a complete phase transition
from ferromagnetic to paramagnetic behavior at 97°C (370 K), which obviously
does not fall into our target temperature range. From Fig 1 one can see clearly
a shift towards the target
temperatures for the grinded and ball-milled powders.
The coarse sand-grinded powder (particle diameter < 150 |im) shows a Curie
temperature of about 72 °C (345 K) and the fine ball-milled powder (effective
particle diameter: 436 nm) shows a temperature of 46°C - 47°C (319 K - 320 K).
Curie temperature is also related with the lattice constants. It has been
reported [15] that with an increase in lattice constant the Curie temperature
increases. Based on the electron diffraction pattern (Fig 2) and the wide angle
X-ray pattern (Fig 3), the d-spacing value (between the 111 planes) has been
calculated and the result has been shown in Table 1. It has been found that the
d-spacing changes from 2.045 in the sand-grinded bulk alloy to 2.036 in the
ball-milled Cu-Ni alloy particles. This might be the reason for decrease in the
Tc value in ball-milled powder. By ball milling nanostructure is obtained by
repeated mechanical deformation by using a number of milling balls and the
internal strain in the crystalline structure caused the change in the
d-spacing. Fig. 4 shows the hysteresis plot of the ball-milled fine powder.
There is no remanent magnetic moment at room temperature indicating the
superparamagnetic [16] behavior of Cu-Ni ball milled powder.
Transmission electron microscopy (TEM) analysis and
particle size analysis verify that sub-micron particles are obtained by this
mechanical alloying method. The particle size was measured after
ultrasonication of an aqueous dispersion for a few minutes and resulted in an
effective diameter of 436 nm with a half width distribution of 218 nm. The
preceding ultrasonic step is essential for particle size measurements, since
metallic nanoparticles tend to agglomerate within a liquid medium.
Figure 5(a),5(b),5(c) was taken from an ultrasonicated
and highly diluted dispersion. The particles that were found during TEM
analysis (using a JEM-2010, JEOL) ranged from around 100 nm to a few
micrometers. The population of spherical particles (Fig. 5(a), ~100 nm) was
very low compared to particles with flake-like geometry (Fig. 5(b)). In these
flake-like particles sub micron grains and boundaries are also observed along
with little contrast. Since copper and nickel have almost the same density
(Cu 8920 kg/m3, Ni 8908 kg/m3) and similar face-centered
cubic lattices with nearly the same lattice constants, changes in contrasts
are only due to different particle thicknesses. The texture of the particles
reflects clearly the abrasive nature of the ball milling process. Fig. 5(c) is
a highly magnified (x 200000) micrograph of the solid texture of a single
(micron sized) particle showing clearly a polycrystalline structure with its
grains and boundaries. The grains have sub micron dimensions.

Figure 5
(a) Cu-Ni nanosized particles (spherical)
after ball milling for 7 days. (b) Cu-Ni nanosized particles (flake-like) after
ball milling for 7 days. (c) High magnification (200000x) TEM micrograph of a
Cu-Ni particle showing its polymorph structure.
Conclusions
Self regulating magnetic hyperthermia can be achieved
by synthesizing nanomagnetic particles with desired Curie temperature. The
desired range of Curie temperatures can be obtained by varying the weight
percentage of nickel and copper based on the phase diagram. Generating particles
in sub micron size is extremely important to vary the Curie temperature. Ball
milling leads to generate submi-cron particles very effectively. Combination of
melting and ball milling is also effective to produce alloy particles in large
quantity; hence it is a commercially viable process. However to control the
monodispersity of the magnetic components and the uniformity in their
distribution throughout the matrix is not very easy. But this non-equilibrium
process has the advantage of generating sub-micron alloy particles easily that
might not be possible using other techniques. The biocompatibility and toxicity
of the produced Ni-Cu nanoparticles have not been investigated in this study.
However, biocompatible coatings could be used to minimize the metallic
interaction with biological structures.
Authors' contributions
Mr. Martin Bettge (a research assistant) and Dr. Jhunu
Chatterjee (an assistant scientist) at the Center for Nanomagnetics and
Biotechnology worked under the supervision of Dr. Yousef Haik. The three
authors contributed to this work equally. All authors read and approved the
final format of the paper.
Acknowledgement
This work has been supported by the Center of
Excellence grant from the Florida State University Research Foundation.
References
- Pankhurst QA,
Connolly J, Jones SK, Dobsen JJ: Applications of magnetic nanoparticles in
biomedicine. Phys D: Appl Phys 2003, 36:R167-R181.
- 2. Kuznetsov AA,
Shlyakhtin OA, Brusnetov NA, Kuznetsov OA: Smart mediators for
self-controlled inductive heating. Eur Cells Mater 2002, 3:75-77.
- Jordan A,
Scholz R, Wurst P, Faehling H, Felix R: Magnetic fluid hyperthermia (MFH):
cancer treatment with AC magnetic field
induced excitation of
biocompatible superparamag-netic nanoparticles. J Magn Magn Mater 1999,
201:41 3-419.
- Chakrabarti
DJ, Laughlin DE, Chen SW, Chang YA: Binary alloy phase diagrams. In
Materials Park Edited by: Massalski TB, Okamoto H, Subramanian PR,
Kacprzak L. Ohio; 1990.
- Lilly MB,
Brezovich IA, Atkinson WJ: Hyperthermia induction with thermally
self-regulated ferromagnetic implants. Radiology 1985, 154:243-244.
- Childress JR,
Chien CL: Granular Fe in a metallic matrix. Appl Phys Lett 1990,56:95-97.
- Koch CC: The synthesis and structure of
nanocrystalline materials produced by mechanical attrition: A review.
Nanos-truc Mater 1993,2:109-129.
- Koch CC:
Materials synthesis by mechanical alloying. Annu Rev Mater Sci 1989,
19:121-143.
- Guo YG, Wan
LJ, Gong JR, Bai CL: Preparation and dispersion of Ni-Cu composite nanoparticles.
Phys Chem Chem Phys 2002, 4:3422-3424.
- Herzer GJ:
Nanocrystalline soft magnetic materials. J Magn Magn Mater 1992, 1 1
2:258-262.
- I 1. Natter
H, Schmelzer M, Hempelmann R: Nanocrystalline nickel and
nickel-copper-alloys: Synthesis, characterization and thermal stability.J
Mater Res 1998, 13:1 186-1 197.
- Leslie-Pelecky
DL, Rieke RD: Magnetic properties of nanostruc-tured materials. Chem Mater
1996, 8:1770-1783.
- Koch CC:
Top-down synthesis of nanostructured materials: Mechanical and thermal
processing methods. RevAdvMater Sci 2003,5:91-99.
- Brady GS,
Clauser HR, Vaccori JA: Materials Handbook 15th edition. Mc Graw-Hill
Professional; 2002.
- Gilleo MA: In
Ferromagnetic Materials Edited by: Wohlfharth EP. North Holland Physics
Publishing; 1986:34.
- Vassiliou JK,
Mehrotra V, Russell MW, Giannelis EP, McMichael RD, Shull RD, Ziolo RF: Magnetic and
optical properties of γ-Fe2O3nanocrystals.
J Appl Phys 1993, 73:5109-51 16.